 
The manufacturer defines framework criteria for risk acceptability in the form of estimated usage, severity of harm and probability of occurrence (para.  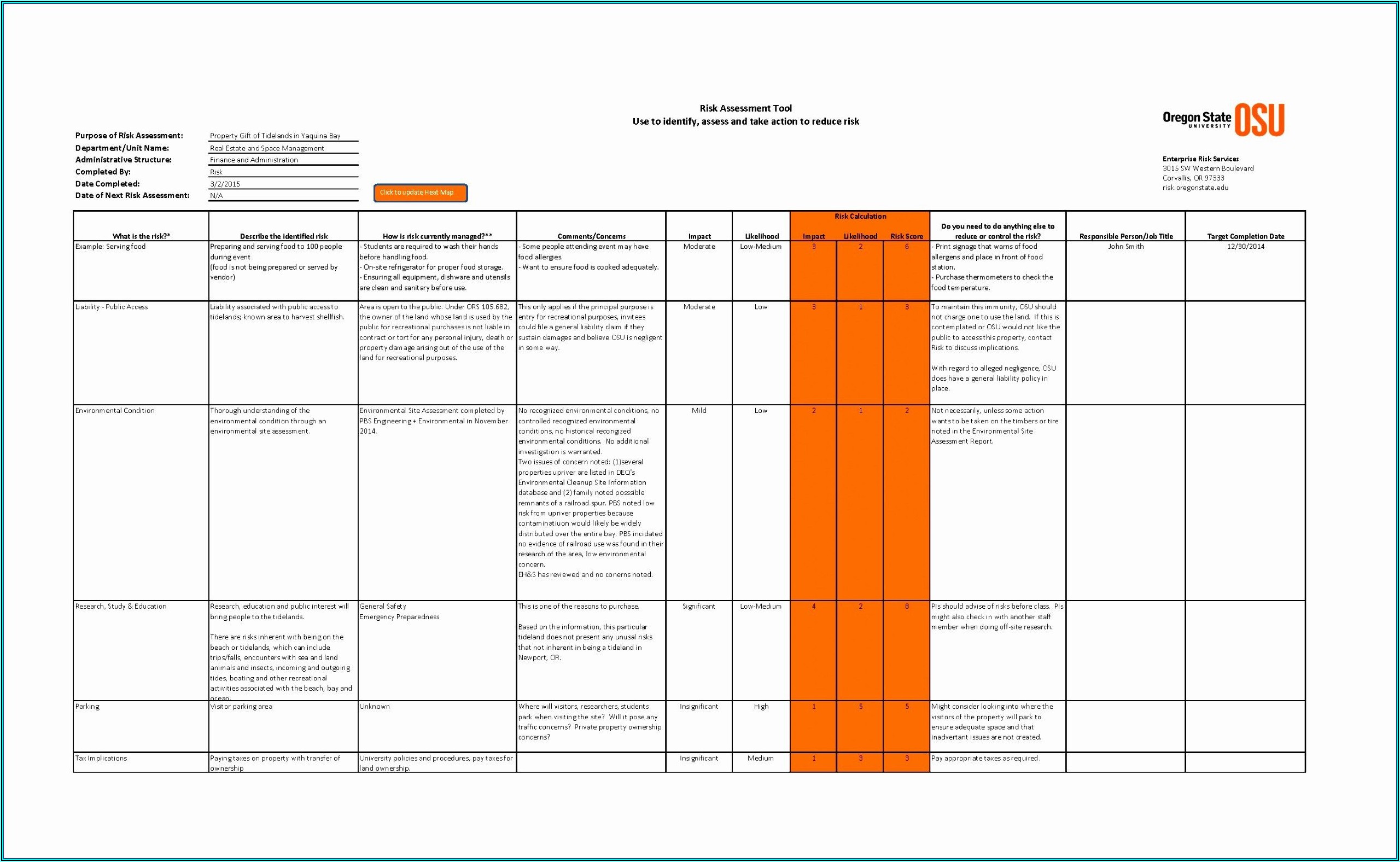
It applies to all people and activities involved in the design, development and distribution process of the medical device, and intends to ensure highest levels of medical device safety consistent with stakeholder expectations. The following policy establishes criteria for risk acceptability following ISO 14971:2019 and ISO/TR 24971:2020. Risk Management Activities are integrated in the software development lifecycle as described in SOP Integrated Relevant Processes 1.1 Risk Management Process and Activities (Records of competence are kept as Part of QMS)ġ. Mapping of Standard Requirements to Document Sections ISO 14971:2019 Section It also references relevant processes and activities which will be conducted for product-specific risk management as part of the integrated software development process (SOP Integrated Software Development). The Risk Management Plan contains the risk policy and defines the criteria for risk acceptance. Berlin Competent Authority (LaGeSo), 06/2022 Risk Management Plan 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
